Assure® Titanium Online Learning
GETTING STARTED
Welcome to the Assure® Titanium Blood Glucose Monitoring System
Thank you for choosing the Assure® Titanium Blood Glucose Monitoring System. The system provides Point-of-Care operators with safe, fast and convenient blood glucose in vitro (i.e., outside the body) diagnostic monitoring. Accurate results can be obtained in just seven seconds with a small (0.5 μL) blood sample. Assure® Titanium is a CLIA waived test and a Certificate of Waiver is required to perform the test in a waived setting.
Laboratories with a Certificate of Waiver must follow the manufacturer’s instructions for performing the test.
Intended Use
- The Assure Titanium Blood Glucose Monitoring System consists of the Assure Titanium Blood Glucose meter and the Assure Titanium Blood Glucose test strips. The Assure Titanium Blood Glucose Monitoring System is intended for use in the quantitative measurement of glucose in fresh capillary whole blood samples drawn from the fingertips. The system is intended for in vitro diagnostic, point of care use in endocrinology clinics and nursing or skilled nursing facilities, for multiple patient use. This system should only be used with single-use, auto-disabling lancing devices for drawing finger stick capillary blood.
- The system is not intended for the screening or diagnosis of diabetes mellitus but is indicated for use in determining dysglycemia.
- The system is not intended for use in acute care or hospital settings.
- The system is not intended for neonatal use.
- The system is for prescription use only.
Assure Titanium Online Learning Course Agenda
- Identify Assure Titanium Meter Features
- Conducting a Blood Glucose Test
- Conducting Control Solution Tests
- Meter Set up
- Cleaning & Disinfecting the Assure Titanium Meter
- Troubleshooting
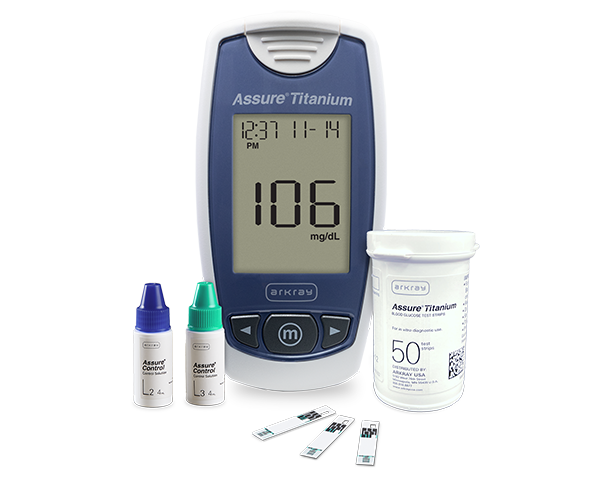
Assure Titanium meter, Assure Titanium test strips and Assure Control Control Solutions are required to complete this course.
